|
11/21/2023 0 Comments Copd flutter device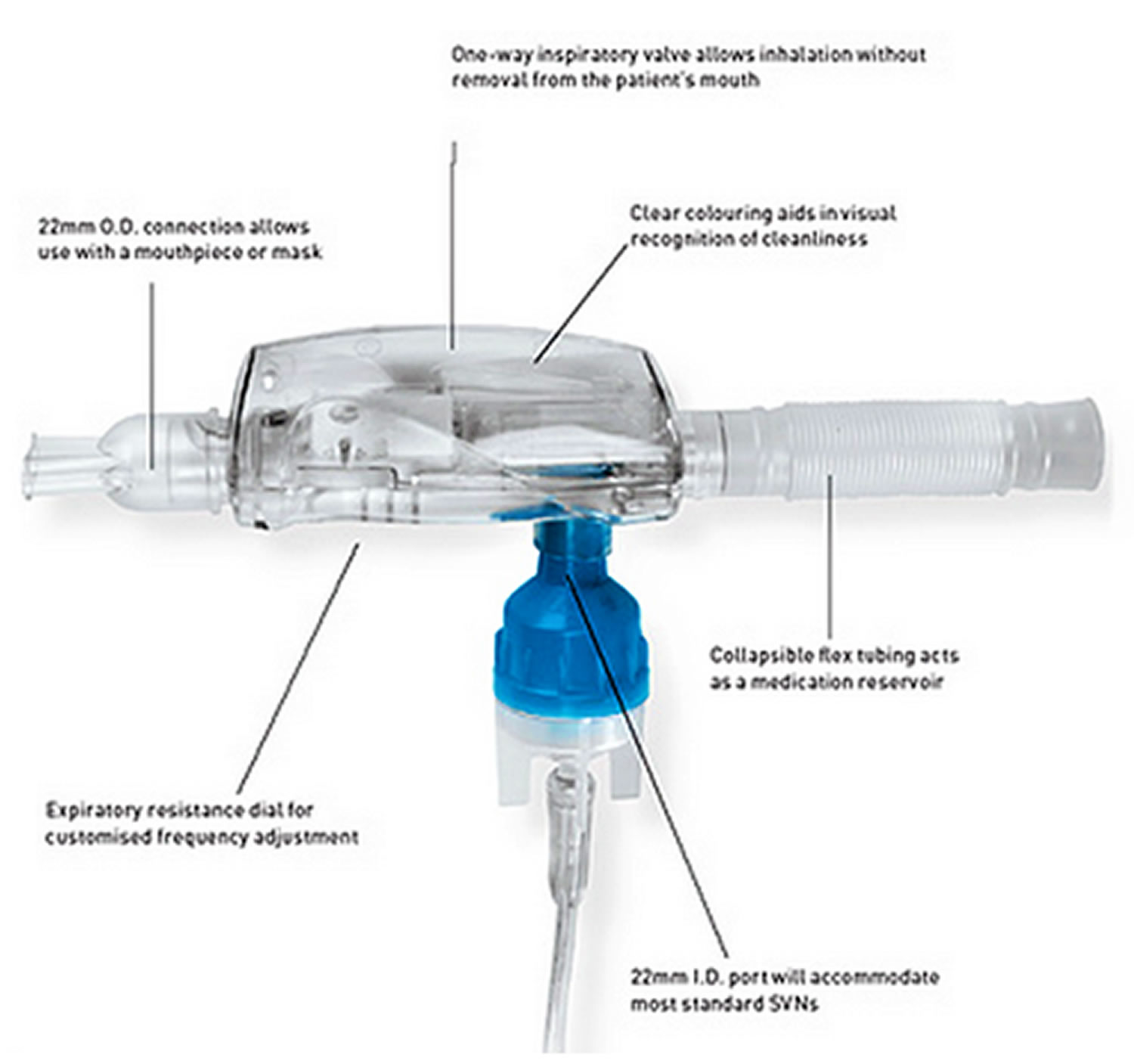
OPEP was designed to promote airway clearance by reducing the viscoelastic properties of the mucus, while splinting open collapsed airways and increasing intrathoracic pressure distal to mucus plugging through collateral ventilation via the canals of Lambert and pores of Kohn. Oscillating positive expiratory pressure (OPEP) devices are handheld, non-pharmacological adjuncts to chest physiotherapy. Airway clearance and physiotherapy have been mainstays in treating hypersecretion since the 1950s, with the first description in the literature purporting the benefits published in 1901. Exacerbations are associated with lower quality of life, more rapid decline in lung function, and higher all-cause mortality. Severe cases can require hospitalisation. 
Patients are prone to exacerbations characterised by periods of acute worsening of symptoms such as dyspnoea, cough, airway inflammation, and excess sputum production (hypersecretion). COPD is the fourth largest cause of death. This response is caused by prolonged exposure to noxious particles or gasses, most commonly cigarette smoke, occupational dusts (from stone cutting, mining, metal grinding etc.) or other environmental dusts (e.g., woodworking). Registration The study was approved as a Clinical Investigation by the Irish Health Products Regulatory Authority (CRN-2209025-CI0085).Ĭhronic obstructive pulmonary disease (COPD) is characterised by non-reversible airflow limitation due to an abnormal inflammatory response in the lungs. Larger, longer duration studies will be required to evaluate efficacy. The device did not negatively impact patients’ lung function, exercise tolerance, or quality of life during short term use (1 month), and usability feedback received was generally positive. No adverse events were recorded during the study or in the follow-up period of 2 weeks. The results indicate that the UL-OPEP is safe to use in patients with COPD. Lung function, 6-min walk test, and SGRQ scores showed no significant change post-study. Overall median age was 67.5 years, range 53–85 years. ResultsĢ4 Participants completed the study: 9 were female. George’s Respiratory Questionnaire (SGRQ), and user experience of the prototype device evaluated using a post-study questionnaire. Quality of life was assessed using the St. Pre- and post-study lung function was assessed with standard spirometry, and exercise tolerance with the 6-min-walk-test (6MWT). Participants were invited to use a prototype disposable OPEP device daily for a period of 1 month. The patients were clinically stable, productive, and not current or previous users of OPEP devices. MethodsĪ sample of 50 volunteer participants were recruited from pulmonary rehabilitation clinics within the local hospital network. Data on user experience of the device were also collected during post-study follow-up. The aim of the current study was to initially evaluate the safety of the prototype in patients with COPD over a period of 1 month to ensure no adverse events, negative impacts on lung function, exercise tolerance, or quality of life. The device was previously evaluated successfully in a group of paediatric CF patients. Adherence to cleaning regimens for respiratory devices is often poor and in response to this, a prototype disposable OPEP device-the ‘UL-OPEP’ (University of Limerick-Oscillating Positive Expiratory Pressure device)-was developed to mitigate the risk of contamination by pathogens. 
Current devices are reusable and require regular cleaning and disinfection to prevent harbouring potentially pathogenic organisms. Handheld oscillating positive expiratory pressure (OPEP) devices have been a mainstay of treatment for patients with hypersecretory conditions such as cystic fibrosis (CF) and chronic obstructive pulmonary disease (COPD) since the 1970s.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
